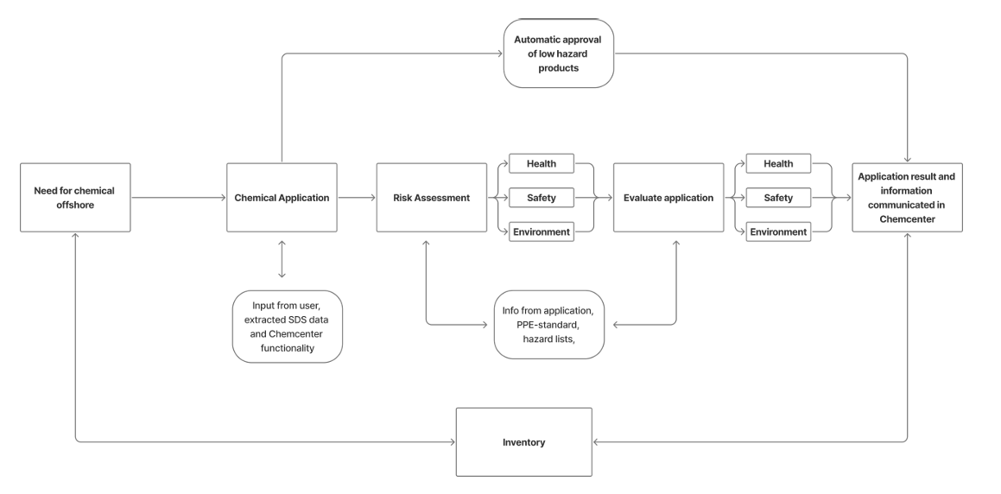
Stages of Chemical Application Approval
Stages of application approval
Create and submit application
The first step is to create and submit a new chemical application request. In this stage, all relevant details about the chemical being requested must be provided by the applicant. The required details include the following:
- Product information – Products are added to an application as safety data sheets. The location, reason for applying, and usage category of the products are included as part of product information.
- Substitution information – Details if the product may or may not be replaced by another that’s less toxic.
- Usage and exposure details – Outlines how the product will be used and the potential exposure level of personnel who will handle the product.
- PPE and barriers definition – Lists and describes the PPE and barriers required to handle the chemical safely. The degree of risk reduced by using the PPE and barriers must also be included.
Low hazard products
After submission of the application, the information provided by the applicant is first automatically assessed in ChemCenter based on a set of criteria specifically defined by your organization. It’s then assigned with an initial risk level. If the product is determined to be low hazard, it may be automatically approved for use without manual review.
Automatic approval may also be triggered if the chemical is classified as PLONOR (Pose Little Or No Risk).
Risk assessment
After submission, the application is sent to someone in your organization who’s responsible for assessing the products for risk to health, safety, and environment. The information provided by the applicant will be used for this assessment. The risks and barriers associated with the chemical are then attached to the application as a result of this assessment.
Application evaluation
The application evaluation stage is where the products in the application is finally approved or declined for use based on the information provided in previous stages. Additional information may still be requested from the applicant if necessary. The HOCNF level for each product is also assigned at this point. This indicates the chemical’s environmental risk level according to the criteria set by national authorities.
Evaluation result
Each product in the application is evaluated separately. The evaluation result for each product is separate from the other products in the application. Some products may be approved, and some may be declined.
When a product is approved for use, it’s added to the Substance Index which is the inventory system in ChemCenter. Approved products have a validity period which can either have a Start Date and an End Date, or be open-ended with a Start Data but no End Date. This validity period may be extended as needed.